Research explainer: Mis-segregation of human chromosomes
Dr Sarah McClelland from Barts Cancer Institute, Queen Mary University of London, has recently published new research in the journal Cell Reports revealing new insights into why cell division can sometimes go wrong.
In this Q & A, she explains why chromosomes can end up in the wrong places when dividing and highlights two particular chromosomes that seem to be more prone to the issue than others.
What is new about the study?

Chromosome mis-segregation during compromised cell division can lead to aneuploidy - the presence of an abnormal number of chromosomes in a cell. But the likelihood of mis-segregation isn't the same for all chromosomes. We show chromosomes 1 and 2 can get stuck more readily than other chromosomes during mitosis, which means they are more likely to become aneuploid.
How did you carry out the study?
We used the new ImageStream cytometer to analyse how individual chromosomes behave when mitosis is disrupted using drugs. We also collaborated to use state-of-the-art single cell sequencing to confirm our findings.
We also used analysis of cells growing on coverslips and carefully analysed with microscopy to catch chromosomes in the act of mis-segregating. We analysed key findings in a panel of different cell types to ensure our findings are relevant to human cell lines in general.
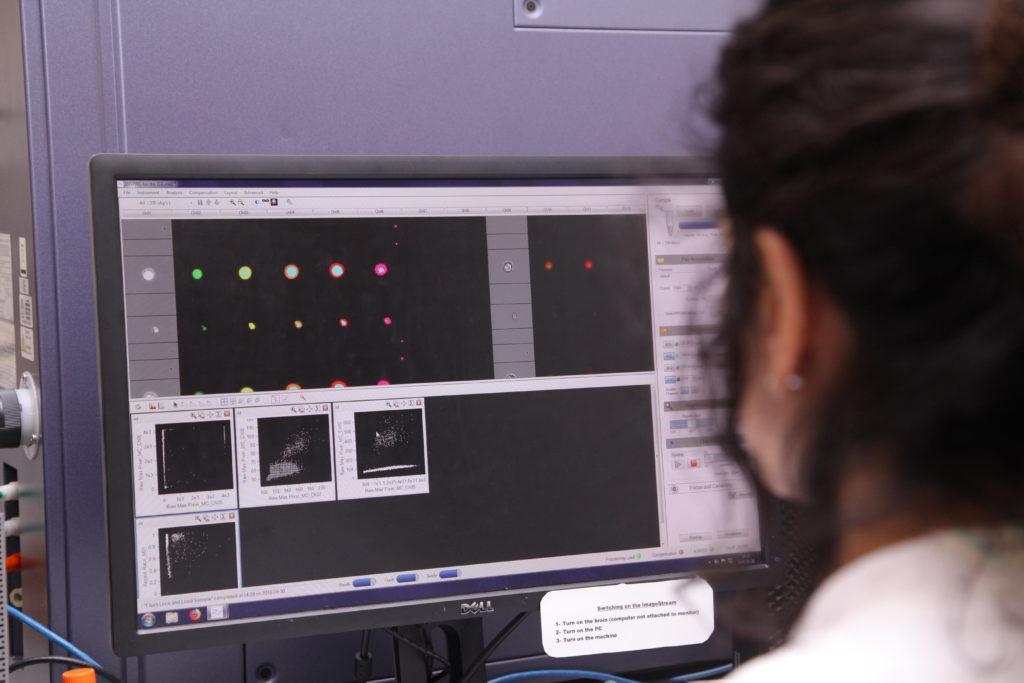
Did you find anything surprising?
The two longest chromosomes (chromosomes 1 and 2) are particularly prone to mis-segregation BUT it is not likely due to their length – at least not in a direct manner. Instead, we discovered that these two chromosomes are susceptible to a gradual weakening of the force that keeps newly replicated chromosomes held together – cohesion.
This means that during drug treatment these chromosomes become less able to attach correctly to the mitotic spindle, a cage that orchestrates accurate division of chromosomes into new cells.
Why is the study important?
This provides the first clear demonstration that human chromosomes are fundamentally different to one another in aspects of chromosome biology that were hitherto unappreciated. It also shows that the development of aneuploidy during cancer might be influenced by these non-random effects.
What are the wider implications of this research?
We tested two mechanisms to disrupt chromosome segregation. However it is likely that different pathways to aneuploidy will affect different chromosomes (in fact our ongoing research also supports this idea). This means there is a very wide scope for further characterising the behaviour of chromosomes in an individual, or personalised manner.
In this way we can discover additional new facets of chromosome biology that can impact the integrity of the human genome during cell growth and division, and begin to integrate these parameters into cancer genomic studies.
The research was conducted in collaboration with the groups of Floris Foijer at the European Research Institute for the Biology of Ageing, University of Groningen, the Netherlands and Elina Vladimirou at the UCL Cancer Institute. The work was majoritively funded by the MRC (PhD studentship), Barts and the London Charity and Wellbeing of Women.
Research paper: ‘Non-random Mis-segregation of Human Chromosomes’ by Joseph Thomas Worrall, Naoka Tamura, Alice Mazzagatti, Nadeem Shaikh, Tineke van Lingen, Bjorn Bakker, Diana Carolina Johanna Spierings, Elina Vladimirou, Floris Foijer, and Sarah Elizabeth McClelland. Cell Reports. doi: 10.1016/j.celrep.2018.05.047
Category: General News, Publications
No comments yet